Aldeyra Therapeutics Achieves Primary Endpoint in Phase 3 TRANQUILITY‑2 Trial in Dry Eye Disease and Intends to Submit New Drug Application for Symptoms and Three Sign Endpoints of Dry Eye Disease
-
Reproxalap Statistically Superior to Vehicle for Both Prespecified Primary Endpoints of
Schirmer Test (p=0.0001) and ≥10 mm Schirmer Test Responder Proportions (p<0.0001) - TRANQUILITY-2 Results May Allow for the Most Comprehensive Dry Eye Disease New Drug Application (NDA) Submission to Date
-
Pending Pre-NDA Meeting with the
U.S. Food and Drug Administration (FDA), Clinical Efficacy Trials of Reproxalap Believed to Be Complete -
Company to Host Conference Call at
8:00 a.m. ET Today
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20220607006390/en/
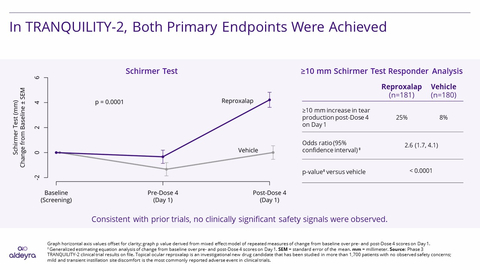
“Schirmer test is an accepted method for measuring tear production and has been used in clinical studies for over 20 years,” said
Pending discussions with the FDA, Aldeyra intends to submit an NDA with ocular dryness symptom score, ocular redness, Schirmer test, and ≥10 mm Schirmer test responder analysis, encompassing results across five adequate and well-controlled completed clinical trials. The submission could represent the most comprehensive NDA submission in dry eye disease to date and allows for the potential of reproxalap to be the first dry eye disease drug approved with symptoms and at least two labeled objective signs. The clinical package is expected to offer unparalleled breadth across acute trials over one to two days of dosing and chronic trials over 12 weeks of dosing, as well as a combination of challenge and field-based assessments.
“Many of my dry eye disease patients complain that current treatments take too long to work, prolonging symptoms and negatively affecting quality of life,” said
A Type B Pre-NDA meeting is expected to be held with the FDA in the third quarter of 2022, followed by a potential NDA submission, pending enrollment in the ongoing 12-month safety trial, and results from a dry eye chamber crossover trial. Enrollment of the crossover dry eye disease trial is substantially complete, and results are expected in the third quarter of 2022. Pending the results, the crossover trial is intended to be submitted to the NDA as a supportive trial.
“The positive results of TRANQUILITY-2 are expected to complete the most comprehensive dry eye disease NDA submission to date,” stated
Conference Call Information
Aldeyra will host a conference call to discuss this announcement at
After the live webcast, the event will remain archived on the Aldeyra website for 90 days.
About Reproxalap
Reproxalap is a first-in-class small-molecule modulator of RASP (reactive aldehyde species), which are elevated in ocular and systemic inflammatory disease. Reproxalap’s mechanism of action has been supported by the demonstration of statistically significant and clinically relevant activity in multiple physiologically distinct late-phase clinical indications.
About Dry Eye Disease
Dry eye disease is a common inflammatory disease estimated to affect 39 million or more adults in
About Aldeyra
Aldeyra develops innovative therapies designed to treat immune-mediated diseases. Our approach is to discover pharmaceuticals that modulate immunological systems, instead of directly inhibiting or activating single protein targets, with the goal of optimizing multiple pathways at once while minimizing toxicity. Two of our lead product candidates, reproxalap and ADX-629, target pre-cytokine, systems-based mediators of inflammation known as RASP (reactive aldehyde species). Reproxalap is in late-stage clinical trials in patients with dry eye disease and allergic conjunctivitis. ADX-629, an orally administered RASP modulator, is in Phase 2 clinical testing for the treatment of systemic immune-mediated diseases. Our pipeline also includes ADX-2191 (intravitreal methotrexate 0.8%), in development for the prevention of proliferative vitreoretinopathy and the treatment of retinitis pigmentosa and primary vitreoretinal lymphoma. For more information, visit https://www.aldeyra.com/ and follow us on LinkedIn, Facebook, and Twitter.
Safe Harbor Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including, but not limited to, statements regarding the timing and submission of a potential NDA; the anticipated timing of enrollment and results from Aldeyra’s clinical trials; expectations regarding the timing and results of the expected Type B Pre-NDA meeting, including the FDA’s acceptance of Aldeyra’s post-hoc review of data and agreement with Aldeyra’s methods of analyzing data; and Aldeyra’s projected cash runway. Aldeyra intends such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 21E of the Securities Exchange Act of 1934 and the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by terms such as, but not limited to, "may," "might," "will," "objective," "intend," "should," "could," "can," "would," "expect," "believe," "anticipate," "project," "on track," "scheduled," "target," "design," "estimate," "predict," "potential," "aim," "plan" or the negative of these terms, and similar expressions intended to identify forward-looking statements. Such forward-looking statements are based upon current expectations that involve risks, changes in circumstances, assumptions, and uncertainties. Aldeyra is at an early stage of development and may not ever have any products that generate significant revenue. All of Aldeyra's development timelines may be subject to adjustment depending on recruitment rate, regulatory review, preclinical and clinical results, and other factors that could delay the initiation, enrollment or completion of clinical trials. Important factors that could cause actual results to differ materially from those reflected in Aldeyra's forward-looking statements include, among others, the timing of enrollment, commencement and completion of Aldeyra's clinical trials, the timing and success of preclinical studies and clinical trials conducted by Aldeyra and its development partners; updated or refined data based on Aldeyra's continuing or post-hoc review and quality control analysis of clinical data, Aldeyra's ability to design clinical trials with protocols, data analysis methodologies, and endpoints acceptable to applicable regulatory authorities; delay in or failure to obtain regulatory approval of Aldeyra's product candidates; the ability to maintain regulatory approval of Aldeyra's product candidates, and the labeling for any approved products; the risk that prior results, such as signals of safety, activity, or durability of effect, observed from preclinical or clinical trials, will not be replicated or will not continue in ongoing or future studies or clinical trials involving Aldeyra's product candidates in clinical trials focused on the same or on different indications; the risk that the results from earlier clinical trials, portions of clinical trials, or pooled clinical data may not accurately predict results of subsequent trials or the remainder of a clinical trial; the scope, progress, expansion, and costs of developing and commercializing Aldeyra's product candidates; uncertainty as to Aldeyra’s ability to commercialize (alone or with others) and obtain reimbursement for Aldeyra's product candidates following regulatory approval, if any; the size and growth of the potential markets and pricing for Aldeyra's product candidates and the ability to serve those markets; Aldeyra's expectations regarding Aldeyra's expenses and revenue, the sufficiency or use of Aldeyra's cash resources and needs for additional financing; political, economic, legal, social, and health risks, including the COVID-19 pandemic and subsequent public health measures, and war or other military actions, that may affect Aldeyra’s business or the global economy; the rate and degree of market acceptance of any of Aldeyra's product candidates; Aldeyra's expectations regarding competition; Aldeyra's anticipated growth strategies; Aldeyra's ability to attract or retain key personnel; Aldeyra’s limited sales and marketing infrastructure; Aldeyra's ability to establish and maintain development partnerships; Aldeyra’s ability to successfully integrate acquisitions into its business; Aldeyra's expectations regarding federal, state, and foreign regulatory requirements; regulatory developments in
In addition to the risks described above and in Aldeyra's other filings with the
____________________
1 Company estimates and Paulsen AJ, Cruickshanks KJ, Fischer ME, et al. Dry eye in the beaver dam offspring study: prevalence, risk factors, and health-related quality of life. Am J Ophthalmol. 2014;157(4):799-806.
2 Choi W, Lian C, Ying L, Kim GE, You IC, Park SH, Yoon KC. Expression of Lipid Peroxidation Markers in the Tear Film and Ocular Surface of Patients with Non-Sjogren Syndrome: Potential Biomarkers for Dry Eye Disease. Curr
View source version on businesswire.com: https://www.businesswire.com/news/home/20220607006390/en/
Investor & Media:
Tel: (857) 383-2409
ALDX@investorrelations.com
Source: